A new study from Karolinska Institutet, published on May 4, 2026, in the journal JAMA Oncology, shows that low doses of the investigational drug endoxifen reduce breast density to the same extent as the standard treatment tamoxifen, but without causing the same troublesome side effects. Endoxifen is the active metabolite of tamoxifen, meaning it is the compound that tamoxifen is converted into in the body to exert its effect.
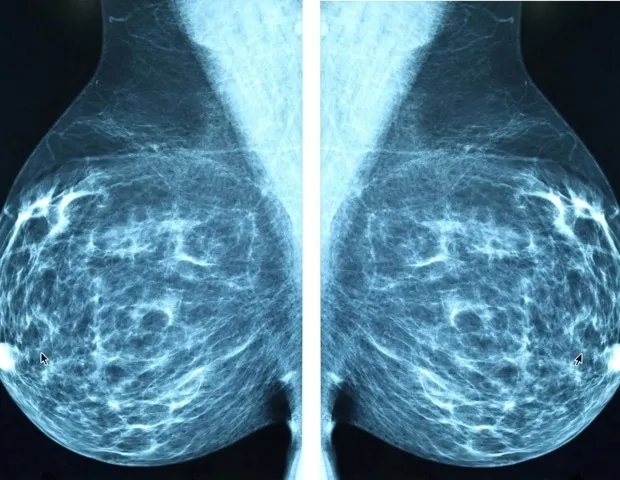
The study, led by researcher Per Hall, involved 240 women at increased risk of breast cancer. Participants were randomly assigned to receive either low-dose endoxifen (2 mg daily) or standard-dose tamoxifen (20 mg daily) for six months. The primary endpoint was change in mammographic breast density, a known risk factor for breast cancer. Results showed that both treatments reduced breast density by approximately 10%, with no significant difference between the groups.
However, side effects differed markedly. Women taking tamoxifen reported higher rates of hot flashes, night sweats, and vaginal discharge, leading to a 30% dropout rate in that group. In contrast, the endoxifen group had a dropout rate of only 10%, with side effects comparable to placebo. The researchers noted that endoxifen's more favorable side effect profile could improve adherence to preventive therapy.
Dr. Hall stated, 'This is the first study to show that low-dose endoxifen is as effective as tamoxifen in reducing breast density, but with significantly fewer side effects. This could make breast cancer prevention more acceptable to high-risk women.' The study was funded by the Swedish Research Council and the Swedish Cancer Society.
Experts caution that larger, longer-term studies are needed to confirm whether the reduction in breast density translates into a lower incidence of breast cancer. Endoxifen is not yet approved by regulatory agencies for breast cancer prevention, but the findings support its potential as a better-tolerated alternative.