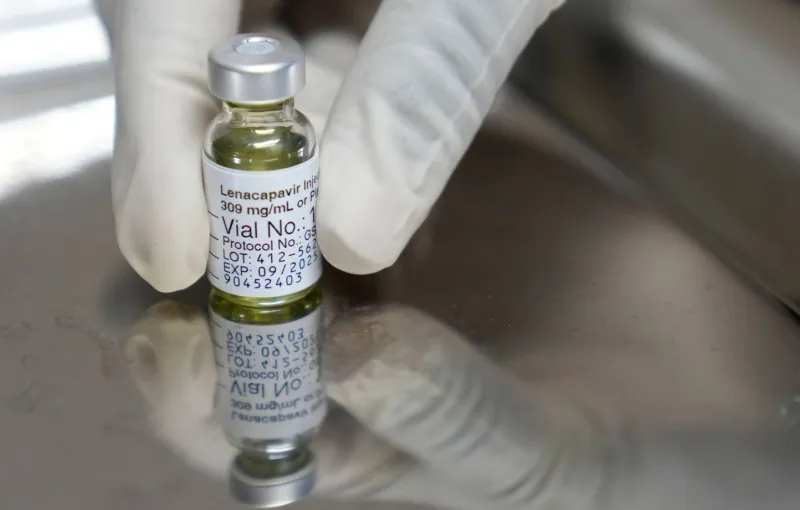
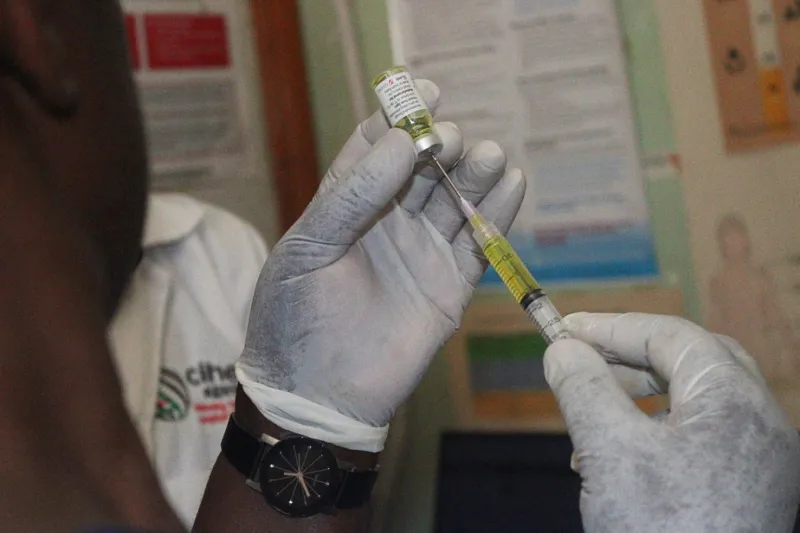
Morocco has been included in an international initiative to expand access to lenacapavir, a long-acting injectable drug for HIV pre-exposure prophylaxis (PrEP). The program is a collaboration between the U.S. President's Emergency Plan for AIDS Relief (PEPFAR) and the Global Fund to Fight AIDS, Tuberculosis and Malaria.
Announced in late 2025, the initiative aims to accelerate the introduction of lenacapavir in up to a dozen countries, primarily in sub-Saharan Africa. The goal is to provide an additional prevention option, particularly for populations at high risk of HIV infection for whom daily oral pills may be a challenge.
Lenacapavir, developed by Gilead Sciences, is administered by injection only twice a year. It received its first regulatory approval for HIV prevention from the European Commission in August 2025 and from the U.S. Food and Drug Administration (FDA) in December 2025. The drug represents a significant advancement in HIV prevention due to its infrequent dosing schedule.
The inclusion of Morocco in this access program is part of a broader effort to make new prevention tools available in diverse regions. The partnership between PEPFAR and the Global Fund will support planning, implementation, and monitoring to facilitate the introduction of the drug in selected countries following local regulatory approvals.