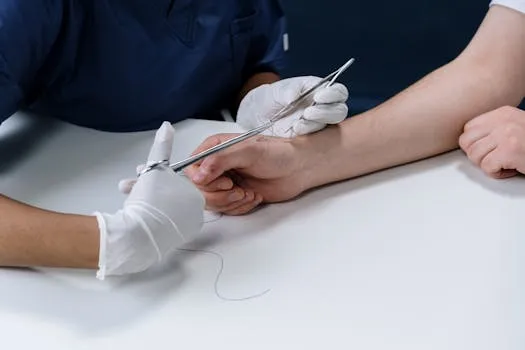
Corza Medical, a global surgical device company, has reaffirmed its commitment to manufacturing surgical gut sutures, a product facing declining demand but essential for certain medical procedures. The company announced on April 28, 2026, that it will continue to produce these sutures to meet the needs of surgeons and patients worldwide.
Gut sutures, made from natural collagen fibers, are used in specific surgeries where absorbable materials are required, such as in ophthalmic and pediatric procedures. Despite market trends favoring synthetic alternatives, Corza Medical emphasizes the unique benefits of gut sutures, including their predictable absorption and handling characteristics.
This decision aligns with Corza Medical's mission to support healthcare providers with a comprehensive portfolio of surgical products. The company operates manufacturing facilities in the United States and Costa Rica, ensuring a stable supply chain for these critical devices.
Industry analysts note that while the overall market for gut sutures is shrinking, niche demand remains strong. Corza Medical's commitment ensures that surgeons have access to these specialized products, preventing potential shortages that could impact patient care.